|
What did solid state NMR tell us? Using a combination of methods, it basically showed us that the clay wasn't actually reinforcing anything. And here's the sad part: it ONLY applies to nylon 6, and not other nylons like the other main commercial one, nylon 6,6. The answer was found using solid state NMR (combined with all the wonderful analyses that had been done by others) to figure this one out. To make a long story short, the answer is complicated because it spans all levels of material organization, from the molecular and nanoscopic to the the micro- and macroscopic. Lots of glass-filled nylons out there with 20-30% glass fiber added to get corresponding modulus improvement, so how does 4% clay accomplish the same thing? What? Doubled? Modulus is a picky property of polymers, very hard to improve even with massive amounts of a reinforcing agent. For example, the heat distortion temperature went up by almost 100 oC and the modulus doubled. 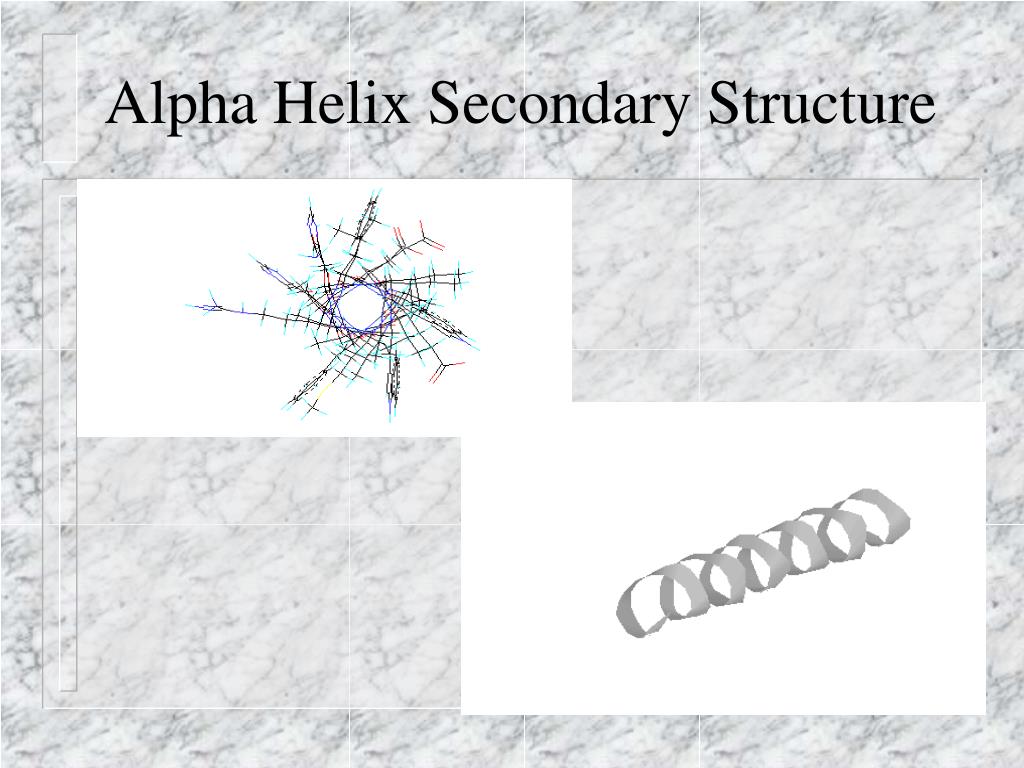
These Japanese researchers in the early 1990s found that even at clay contents of around 3-5%, the properties of nylon 6 were enormously improved. What say we delve into this fantastic discovery in more depth? There's plenty of it and it's really cheap, so why not?" Actually, this works,īut the scientists who discovered it knew what they were doing- it was a special kind of dirt called Wrong! That always works for fairies, but hey, this is science! So you think for a minute and decide, Turns out it's really easy but not at all obvious how to do this: just So one of the big questions facing you (if you happen to be a manufacturer of nylon 6) is how to make your nylon as The other most common commercial nylon, the AA-BB nylon 6,6, is more symmetrical, withĪmide groups formed from 1,6-hexandiamine and the 6-carbon adipic acid. Nylon 6 has a reported Tm of 220 oC or less, depending on what type ofĬrystal structure is present in the material. That is polymerized through a ring-opening process shown below. The amino-acid, but from a cyclic amide monomer, Have the two types of functional groups (carboxylic acid and amine) used to make the amide linkage. This nylon is an A-B polymer, meaning that the monomer and repeat unit both 
It has the 3D structure shown in the figure above. Nylon 6 is one of the most common commercial nylons sold today. Use your mouse on the model to rotate and zoom.Ĭlick items below for other modifications.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
